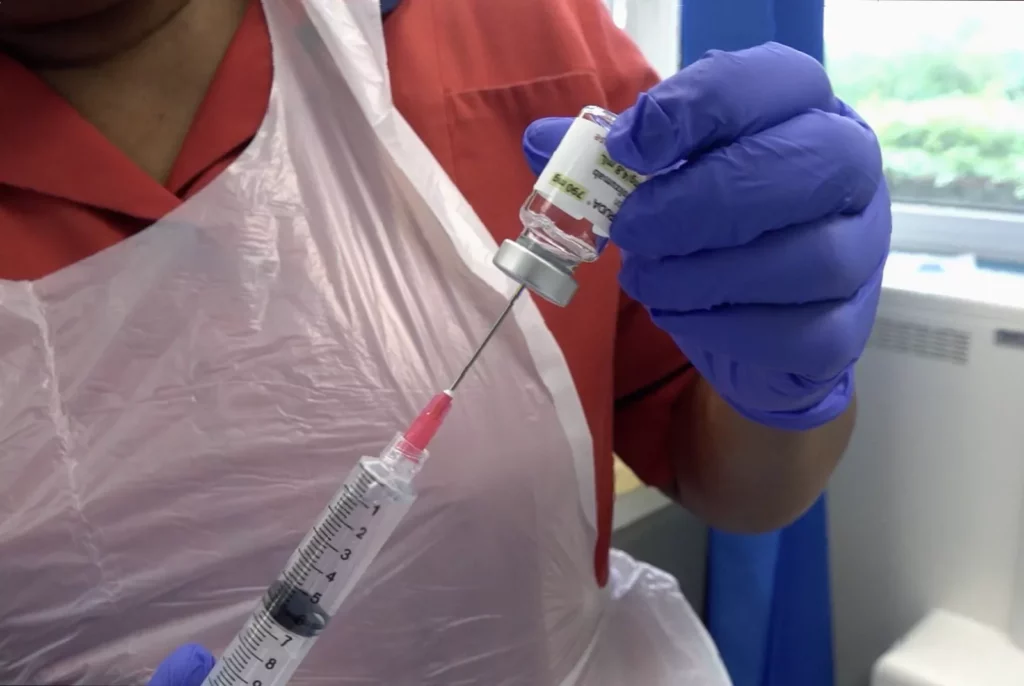
A one-minute cancer jab is now available on the NHS, offering tens of thousands of patients a dramatically faster way to receive immunotherapy treatment for more than a dozen forms of the disease. The subcutaneous injection of pembrolizumab, sold under the brand name Keytruda, is being rolled out across hospitals in England following regulatory approval, with the first patients already treated at the Mount Vernon Cancer Centre in Hertfordshire.
The drug works by blocking a protein called PD-1, which acts as a natural brake on the immune system. By releasing that brake, pembrolizumab enables the body to recognise and attack cancer cells. It is approved for 32 indications across 14 cancer types, including lung, breast, head and neck, and cervical cancers. Around 14,000 patients in England begin pembrolizumab therapy each year, and the majority are now expected to move to the injectable form.
From drip to jab: hours slashed to minutes
The most striking change is in treatment time. Until now, patients had to sit for up to two hours receiving the drug intravenously. The process involved careful preparation of a 100ml bag — roughly a third of a can of Coke — under sterile conditions, plus insertion of a cannula into the vein. Vikash Dodhia, head of pharmacy at Mount Vernon, explained that an IV infusion typically took between 30 and 45 minutes to administer. “The patient also then has to have a cannula inserted into their veins to receive that particular treatment,” he said.
The new formulation is just 4ml — less than a teaspoon — and is injected into the abdomen in one to two minutes. “So for a patient, it means they get all of that time back,” Dodhia added. The total time saving is up to 90 per cent. The treatment is given every three weeks as a one-minute injection, or every six weeks as a two-minute injection, depending on the type of cancer.

Clinical studies have shown that the subcutaneous version offers comparable effectiveness to the IV drip. In a Phase III trial, objective response rates were 45.4 per cent for the injection versus 42.1 per cent for the drip, with a consistent safety profile. The drug itself costs the NHS exactly the same amount.
Patients speak of relief and freedom
Among the first to receive the new injection was Shirley Xerxes, an 89-year-old from St Albans who has bowel cancer. She was treated at the Mount Vernon Cancer Centre, part of East and North Hertfordshire Teaching NHS Trust. “Having the injection has been great and the nurse was so good, so kind,” she said. “I just had it in my tummy. But it’s not as quick as a Covid jab. It takes a couple of minutes. I feel appreciative, really. I mean, we don’t have to pay for it. It’s good, I’m very appreciative. It’s been wonderful. Now I can spend more time on gardening, especially now spring is here.” Dodhia believes Xerxes was the first patient in the country to receive this specific treatment.
Stephen Friend, 67, who has melanoma, was also treated at Mount Vernon. “This new process takes away a lot of the stress, I think,” he said. “Up until now I’ve been extremely healthy, I’ve not really used the NHS up until now. And it’s just been amazing.”

Staff capacity and NHS productivity
Behind the scenes, the switch from intravenous to subcutaneous delivery is also transforming how hospital teams work. Previously, pharmacy staff had to prepare each IV bag under sterile conditions — a time-consuming process. The ready-to-use injection eliminates that step. MSD, the manufacturer of Keytruda, has calculated that the injection cuts the amount of time staff spend on preparation by 44 per cent. The NHS estimates that the nationwide rollout could save more than 100,000 hours of preparation and treatment time each year — the equivalent of around 11 years.
“For staff, if you’re able to give the treatments much quicker, that means you can actually treat more patients through the treatment chairs,” Dodhia said. “So in a unit like this we are able to then accommodate more patients in a given day and actually treat patients quicker. And we all know, particularly in cancer, the sooner you start the treatment, the better benefit there is.”
Professor Peter Johnson, NHS national clinical director for cancer, called the immunotherapy “a lifeline for thousands of patients”. He said: “Managing cancer treatment and regular hospital trips can be really exhausting, and not only will this innovation make therapy much quicker and more convenient for patients, it will help free up vital appointments for NHS teams to treat more people and continue to bring down waiting times.”

James Richardson, national specialty adviser for cancer drugs at NHS England, described the change as a “win-win innovation, because patients will spend far less time in hospital and, crucially, our clinical teams will have more capacity to care for others.” Michelle Mitchell, chief executive of Cancer Research UK, said the jab will “speed up the delivery of treatment, allowing people to spend more time living their lives outside of hospital, as well as freeing up time for staff to care for other patients.”
Health and Social Care Secretary Wes Streeting, a cancer survivor himself, said: “As a cancer survivor, I know how important quick treatment is, and this rollout will offer quicker, more convenient care, saving patients time and helping them in their recovery with less time in hospital. Not only that – it’ll also free up valuable time so clinicians can care for even more people and potentially save even more lives.”
The subcutaneous formulation of pembrolizumab is the first MHRA-approved injectable immune checkpoint inhibitor of its kind. It contains recombinant berahyaluronidase alfa, an enzyme that acts as a permeation enhancer to help the drug absorb into the body. Keytruda is the world’s best-selling prescription medicine, with global sales of $30 billion (£22 billion) in 2025. The NHS had previously introduced injectable forms of other immunotherapy drugs, such as nivolumab, making pembrolizumab the third such drug available as a jab.