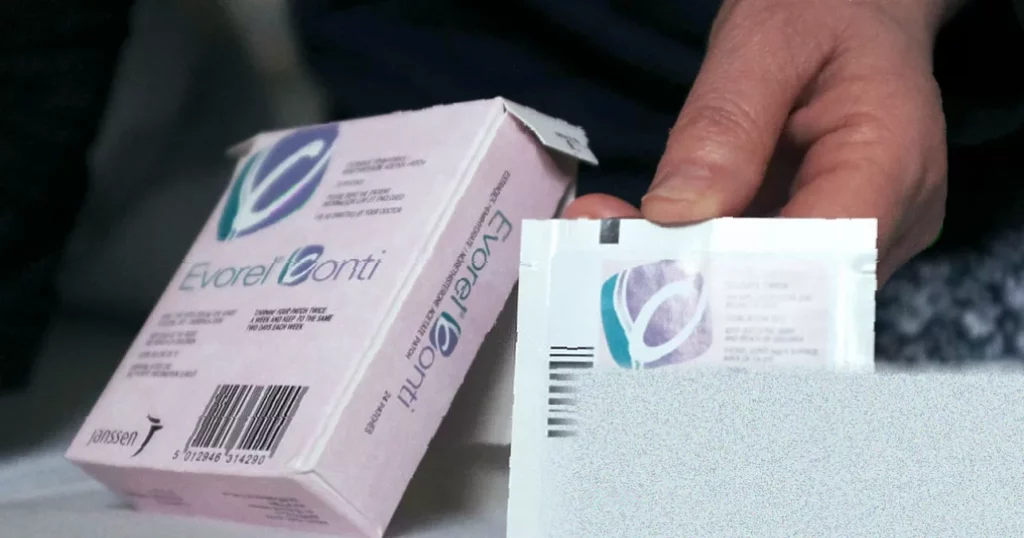
A major hormone replacement therapy producer has been formally censured for “systemic failures” that put patients at risk, following whistleblowing by its own staff. Theramex, the UK company behind widely used HRT products Evorel and Intrarosa, has been issued with a public reprimand by the industry’s self-regulatory body.
A catalogue of compliance failures
The Prescription Medicines Code of Practice Authority (PMCPA) found Theramex in breach of fundamental standards on 21 separate counts. The panel stated these breaches not only jeopardised patient safety but also “brought discredit upon, and reduced confidence in, the pharmaceutical industry”. The failings were highlighted by Theramex employees who raised “alarming” compliance issues internally, alleging their concerns were “brushed off”.
Central to the case was the company’s failure to maintain accurate and up-to-date prescribing information for its medicines. For Intrarosa (prasterone), a local hormone therapy for postmenopausal women, crucial product information had not been updated since 2019. For the Evorel patch, a transdermal HRT containing estradiol, the company failed to provide comprehensive side-effect information. This is particularly significant given the scale of use: Evorel patches are among the most prescribed forms of transdermal HRT, with over 250,000 items issued last financial year, part of nearly 10 million estradiol items prescribed in 2024/25.
In a separate but equally serious breach, Theramex was reprimanded for advertising at a reproduction and advertising conference. The company failed to specify that its uterine fibroid treatment, Yselty (linzagolix), must not be used during pregnancy. The PMCPA noted it was “inconceivable that all attendees would have this knowledge,” especially as the drug’s mechanism of action was not stated. Yselty is contraindicated in pregnancy due to the risk of early pregnancy loss.
Exit from self-regulation condemned
In a move condemned by regulators, Theramex withdrew from the PMCPA’s jurisdiction in January 2026, leaving the UK pharmaceutical industry’s self-regulatory framework. The PMCPA stated this decision “inevitably delayed any regulatory action and oversight” and was a “derogation of responsibility under self-regulation” to public safety. The Medicines and Healthcare products Regulatory Agency (MHRA) will now assume full responsibility for regulating the company in the UK.
Dr Amit Aggarwal, medical director of the Association of the British Pharmaceutical Industry (ABPI), whose code Theramex breached, said the company had “fallen seriously short of the standards expected under our strict ABPI code of practice”. He expressed disappointment at its departure from a system that “holds companies to standards above and beyond the law.”
Julian Beach, the MHRA’s executive director of healthcare quality and access, also voiced disappointment but emphasised that leaving the PMCPA “does not mean a company escapes scrutiny.” He confirmed the MHRA retains legal powers to investigate and act on public safety concerns, noting that breaches of regulations “can amount to criminal offences.”
Theramex, a global specialty pharmaceutical company focused on women’s health which was re-established in 2018 and is owned by private equity firms PAI Partners and The Carlyle Group, defended its actions. A company spokesperson said it acted promptly to address “historical matters,” had undertaken a comprehensive compliance review including an independent audit, and concluded that direct regulation by the MHRA was “most appropriate” while upholding the spirit of industry codes.
This compliance scandal emerges against a backdrop of significant challenges in HRT access in the UK, where shortages have been reported in recent years due to surging demand, manufacturing issues, and supply chain disruptions.