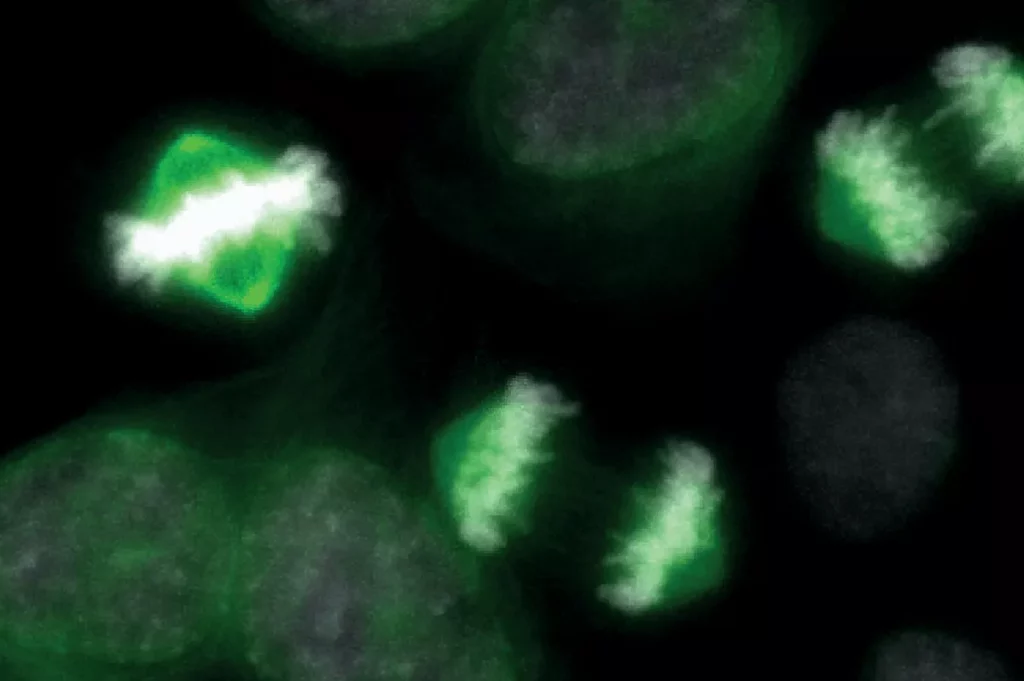
Scientists are studying the patterns of cell division in both healthy and cancerous cells, aiming to understand how the molecular machinery that separates chromosomes can malfunction and drive tumour growth. The research focuses on the kinetochore, a large protein complex that forms at the centre of each chromosome and is responsible for ensuring accurate segregation during mitosis.
Deciphering the Kinetochore’s Role
The work is led by Professor Andrea Musacchio, a director at the Max Planck Institute of Molecular Physiology in Dortmund and a professor at the University of Duisburg-Essen. His group specialises in the biochemical reconstitution of the kinetochore, using structural biology and cell biology to understand its assembly and function. The kinetochore acts as the critical attachment point for spindle microtubules, which pull chromosomes apart, and also plays a central role in the Spindle Assembly Checkpoint (SAC) – a surveillance mechanism that halts cell division if chromosomes are not properly attached. Defects in kinetochore function can lead to aneuploidy, an abnormal number of chromosomes that is a hallmark of many cancers.
Recently, Musacchio’s group, in collaboration with Stefan Raunser, has elucidated the structural organisation of the kinetochore corona, the outermost layer of the complex, and its assembly mechanism. This detailed understanding of normal kinetochore architecture provides a baseline for studying what goes wrong in malignant cells.
How Cancer Cells Evolve and Resist Chemotherapy
Central to the research is the question of how cancer cells exploit errors in cell division to evolve and become resistant to chemotherapy. Malfunctioning kinetochores contribute to chromosomal instability (CIN), a condition in which cells continually gain or lose chromosomes during division. This instability generates genetic diversity within a tumour, allowing subsets of cancer cells to survive treatment and adapt. The uncontrolled proliferation of cancer cells is itself a result of dysregulated cell cycle progression, and the genetic variations arising from segregation errors provide the raw material for resistance.
The team is investigating the molecular “signals” that control these processes. A key area of investigation is the rate at which chemical tags called phosphates are attached to and detached from proteins – known as phosphorylation–dephosphorylation (PdP) dynamics. This rapid cycling acts as an on/off switch for proteins and is believed to govern cell behaviour. By deciphering these hidden signals, the researchers hope to understand how cells divide accurately under normal conditions and how that precision is lost in cancer, opening potential routes to correct the errors or halt cancer cell proliferation.
Professor Musacchio said: “Our expertise in the biochemical reconstitution of the kinetochore complements the diverse skillsets of our team and gives us the opportunity to understand these patterns during cell division in healthy cells, and what goes wrong in cancer cells that allow them to evolve and become resistant to chemotherapy.”
The eight-year project is funded by a £4 million award from Wellcome and is a collaboration between the University of Dundee and the Max Planck Institute of Molecular Physiology. The study of kinetochore-microtubule attachments is considered an appealing therapeutic target, and the research builds on existing approaches in cancer therapy. Mitotic poisons, which target cell division, are already used in the clinic, and cell-cycle-targeting drugs such as CDK4/6 inhibitors have been licensed for clinical use. A broader landscape of innovation includes immunotherapy (checkpoint inhibitors, CAR T-cell therapy), oncolytic virus therapy, and targeted therapies, though these are not directly linked to the kinetochore work. The new research aims to identify precisely how the cellular switches can be manipulated to correct division errors or stop cancer cells from proliferating.