A patient inadvertently received the wrong antidepressant after discovering Citalopram 40mg tablets inside a sealed carton marked as Sertraline 100mg, prompting a precautionary recall by the UK medicines regulator.
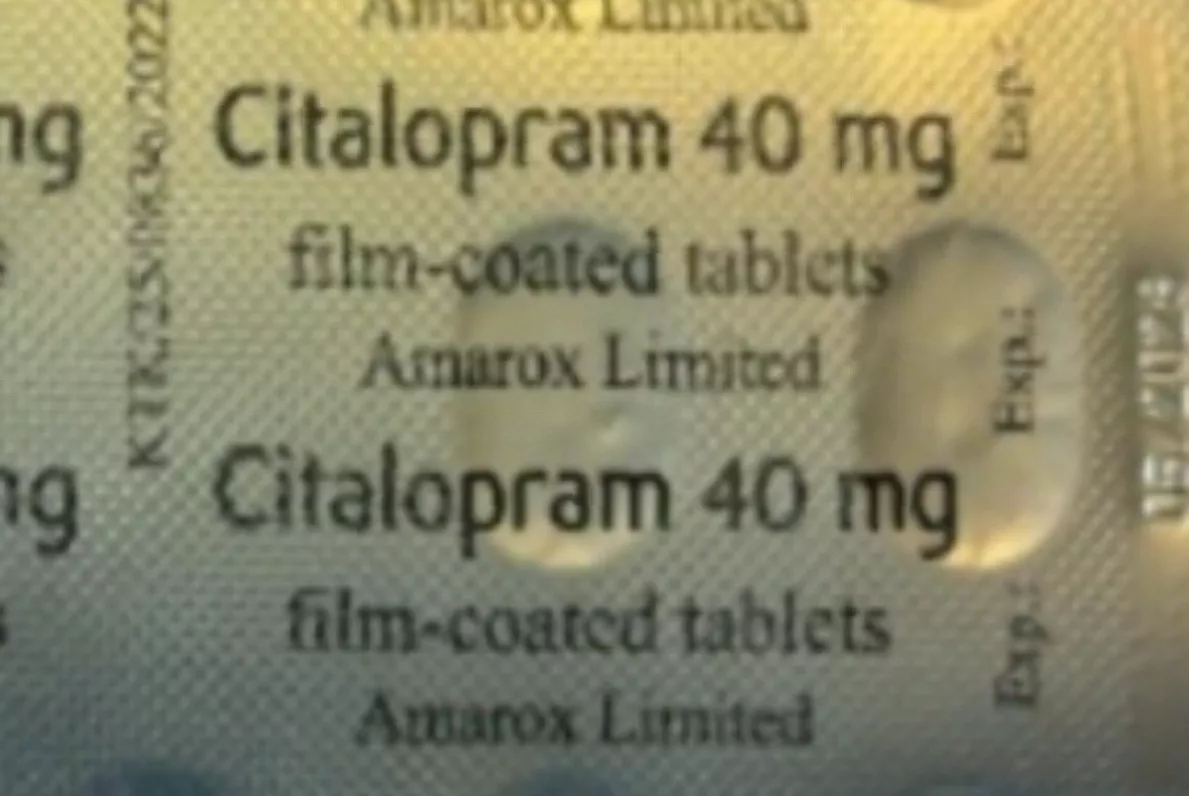
The Medicines and Healthcare products Regulatory Agency (MHRA) has issued a Class 2 recall affecting a single batch – number V2500425 – of Sertraline 100mg film-coated tablets manufactured by Amarox Limited. A Class 2 recall indicates a defect that could cause mistreatment or harm but is not immediately life-threatening, with action required within 48 hours.
Amarox Limited, a UK subsidiary of the Indian pharmaceutical giant Hetero Group established in 2017, produces both sertraline and citalopram at the same manufacturing site. The company became commercially active in the UK in November 2019, specialising in generic medicines for the NHS. The error is believed to have occurred during the secondary packaging process, when blister strips are placed into their outer cartons, resulting in a citalopram blister strip being placed inside a sertraline box.
The mix-up: two SSRIs, one packaging line
Both medications belong to the Selective Serotonin Reuptake Inhibitor (SSRI) class, which works by increasing serotonin levels in the brain to treat depression and anxiety disorders. However, the accidental substitution — or addition — of citalopram alongside sertraline can pose serious health risks. Patients who have mistakenly taken citalopram instead of or in addition to their prescribed sertraline may experience heightened serotonergic effects, including nausea, headaches, disrupted sleep patterns, mild anxiety, diarrhoea, tremor, indigestion, decreased appetite, excessive sweating, and ejaculation failure.
More serious potential complications include serotonin syndrome — a rare but life-threatening condition caused by excessive serotonin accumulation, with symptoms such as agitation, confusion, rapid heart rate, high blood pressure, dilated pupils, muscle rigidity, tremor, and hyperthermia. Both drugs can also affect the electrical activity of the heart, increasing the risk of QT prolongation, a serious heart rhythm problem that can cause fainting or palpitations. Other risks include increased bleeding tendency due to altered platelet function, hyponatremia (low sodium levels), activation of mania or hypomania in individuals with bipolar disorder, seizures, and angle closure glaucoma in predisposed individuals.
Patient checks: how to identify affected medication
Dr Alison Cave, the MHRA’s Chief Safety Officer, has issued clear guidance for anyone prescribed Sertraline 100mg tablets. “If you have been prescribed Sertraline 100mg tablets and have received batch number V2500425, please check that the carton contains the right medication. You can find the batch number and expiry date printed on the side of the outer packaging,” she said.
Patients are instructed to open the carton and examine the blister strips inside. If the strips are labelled “Citalopram 40mg”, they should contact their pharmacy as soon as possible. If the strips are labelled “Sertraline 100mg”, no further action is needed. Dr Cave added that individuals who suspect they have inadvertently taken citalopram — either instead of or alongside their sertraline — or who are experiencing any adverse effects, should seek medical guidance without delay.
The MHRA advises that any suspected adverse reactions should be reported through the Yellow Card scheme, the UK’s spontaneous reporting system for adverse drug reactions, which can be used by both healthcare professionals and members of the public via the website, app, or post.
Directions for pharmacies and healthcare professionals
Healthcare professionals have been directed to cease supplying the affected batch immediately and return all remaining stock to their suppliers. Pharmacists and dispensing staff must identify patients who may have received the compromised product and request its return if any medication remains. Where affected patients are identified, the professional should liaise with the patient’s GP or responsible clinician to arrange a treatment review and determine whether a fresh prescription is necessary.
Particular attention and monitoring are warranted for certain patient groups, including those aged over 65 or under 18, individuals with cardiac or liver conditions, and those whose bodies metabolise medications differently.
Pharmacy staff are reminded to report all medication errors, even if no injury occurred, through the National Reporting and Learning Systems or, if harm has occurred, to file an incident report with the General Pharmaceutical Council. The broader context of packaging errors — including concerns about barcode mislabelling on medicine packs — underscores the critical role of patient vigilance in identifying errors, as demonstrated by the patient who reported the initial mix-up that led to this recall.

